Finance Minister Nirmala Sitharaman’s customs duty wipeout on life-saving oncology medicines is making headlines — but the real story is in the details patients actually care about.
India has roughly 2.5 million new cancer cases every year. For most of those patients, the fight isn’t just biological — it’s financial. Advanced targeted therapies, the kind that have genuinely transformed survival odds over the past decade, are almost entirely imported. And every single one of them carried a customs duty surcharge that was quietly, reliably making an already devastating illness a little more expensive to survive.
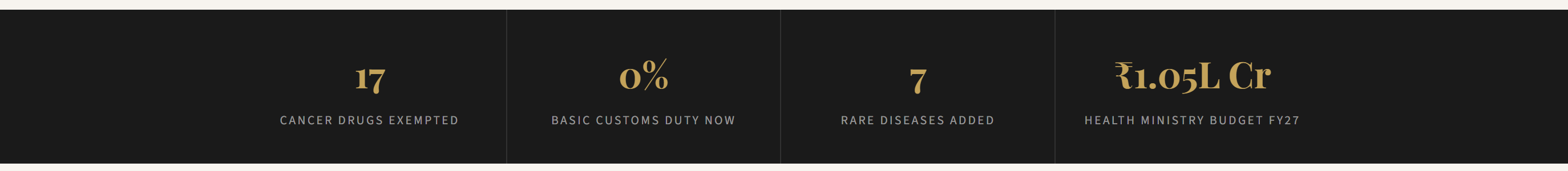
On February 1, presenting the Union Budget 2026-27 in Parliament, Finance Minister Nirmala Sitharaman announced a full exemption of basic customs duty on 17 cancer drugs. Not a reduction. Not a partial rebate. A complete removal — from a previous rate of 5 to 10 per cent down to zero, with immediate effect. Alongside that, seven more rare diseases were brought under the duty-free umbrella for personal imports of medicines and Food for Special Medical Purposes.
It’s a policy move that pharma analysts are calling “modest but meaningful.” For the patients sitting in oncology wards across the country, the language is simpler: something that was broken just got fixed — at least a little.
“The exemption of basic customs duty on 17 drugs used in cancer treatment is one of the most impactful measures. This will provide much-needed financial relief to patients and their families.”
Why a Customs Duty on Medicine Even Exists
This is a question worth pausing on, because it trips up a lot of people. India does not manufacture most advanced cancer drugs. The complex biologics — the targeted therapies, the immunotherapies, the CAR-T cell treatments — are produced overseas by companies like Novartis, Eli Lilly, AstraZeneca, and Roche. When those drugs enter the country, they pass through customs. And customs, like everything else in a government’s revenue toolkit, charges a fee.
That fee was sitting between 5 and 10 per cent on these particular drugs. On paper, that doesn’t sound like much. In practice, when a single monthly course of a CDK4/6 inhibitor costs several lakhs, even a 5 per cent duty adds up to tens of thousands of rupees — every single month, for years of treatment. For a one-time CAR-T cell therapy priced at ₹25–30 lakh, the duty was adding lakhs on top of an already staggering bill.
Manufacturers and distributors typically pass import costs directly to patients, especially in private hospitals. So the customs duty wasn’t just a line item on a government ledger. It was an invisible tax on people who were already spending everything they had to stay alive.
The Complete List: All 17 Drugs, Now Duty-Free
The government didn’t release these names with fanfare. They were listed in the Budget documents and a gazette notification. Here is the full list, along with what each drug is actually used to treat — because that context matters more than the policy language.
Complete List — 17 Cancer Drugs Exempted from Basic Customs Duty
| # | Drug Name | Primary Use / Cancer Type | Status |
|---|---|---|---|
| 1 | Ribociclib | Hormone receptor-positive breast cancer (CDK4/6 inhibitor) | Duty-Free |
| 2 | Abemaciclib | Hormone receptor-positive breast cancer (CDK4/6 inhibitor) | Duty-Free |
| 3 | Talicabtagene autoleucel (NexCAR19) | Relapsed/refractory B-cell cancers (India’s first domestic CAR-T therapy) | Duty-Free |
| 4 | Tremelimumab | Advanced hepatocellular (liver) carcinoma | Duty-Free |
| 5 | Venetoclax | Chronic lymphocytic leukaemia (CLL) | Duty-Free |
| 6 | Ceritinib | ALK-positive non-small cell lung cancer | Duty-Free |
| 7 | Brigatinib | ALK-positive non-small cell lung cancer | Duty-Free |
| 8 | Darolutamide | Castration-resistant prostate cancer | Duty-Free |
| 9 | Toripalimab | Melanoma and other solid tumours (PD-1 checkpoint inhibitor) | Duty-Free |
| 10 | Serplulimab | Extensive-stage small cell lung cancer (PD-1 inhibitor) | Duty-Free |
| 11 | Tislelizumab | Multiple tumour types — gastric, oesophageal, nasopharyngeal cancers | Duty-Free |
| 12 | Inotuzumab ozogamicin | Relapsed/refractory acute lymphoblastic leukaemia (ALL) | Duty-Free |
| 13 | Ponatinib | Chronic myeloid leukaemia (CML) — resistant or intolerant cases | Duty-Free |
| 14 | Ibrutinib | Chronic lymphocytic leukaemia and mantle cell lymphoma | Duty-Free |
| 15 | Dabrafenib | BRAF-mutant melanoma and non-small cell lung cancer | Duty-Free |
| 16 | Trametinib | BRAF-mutant melanoma (used in combination with dabrafenib) | Duty-Free |
| 17 | Ipilimumab | Melanoma, renal cell carcinoma, liver cancer (CTLA-4 checkpoint inhibitor) | Duty-Free |
Look at that list carefully. It’s not random. The government targeted therapies across the broadest range of cancer types — breast, lung, liver, prostate, skin, blood cancers, and leukaemias. These are drugs that oncologists have been prescribing for years, but whose costs were consistently out of reach for a significant share of the patients who needed them.
What This Actually Means for a Patient
Let’s be honest about the gap between policy announcements and what happens inside a hospital. Customs duty removal does not make cancer treatment free. It does not guarantee that every patient will suddenly afford these drugs. Insurance coverage in India remains patchy, and many of these therapies still exceed what most plans will cover.
The Real-World Math
A monthly course of a CDK4/6 inhibitor like Ribociclib can run between ₹1.5 lakh and ₹3 lakh per cycle. At the previous 5–10% duty rate, a patient was effectively paying ₹7,500 to ₹30,000 extra every month — just in import taxes. Over a year of treatment, that’s ₹90,000 to ₹3.6 lakh in pure duty costs, on top of everything else.
For CAR-T therapies like NexCAR19, a single duty charge on a ₹25 lakh treatment could add ₹1.25 to ₹2.5 lakh to the final bill. That’s not a rounding error — that’s a family’s decision point.
What the exemption genuinely does is remove a structural penalty from the system. For patients on long-term therapies — breast cancer patients on CDK4/6 inhibitors, leukaemia patients on venetoclax, those receiving repeated immunotherapy cycles — the cumulative savings over the course of treatment become meaningful. It also improves adherence. When treatment costs drop even slightly, patients are less likely to delay or skip doses. In oncology, that distinction can be the difference between remission and relapse.
The Rare Disease Expansion: Seven New Conditions Covered
Alongside the cancer drugs, the Budget expanded duty-free import coverage to medicines and nutritional supplements for seven additional rare diseases. This matters because rare disease patients in India often have no domestic treatment option at all. Their medications are sourced entirely through personal imports — a process that was, until now, taxed.
The expanded list includes conditions like Primary Hyperoxaluria Type 1 (where patients need repeated doses of lumasiran over a lifetime), cystinosis, and Hereditary Angioedema — a condition where emergency treatments can cost several lakhs per vial. For Primary Immunodeficiency Disease patients who depend on regular intravenous immunoglobulin infusions costing tens of thousands per session, the per-dose savings from zero duty adds up fast.
The rare disease exemption is time-bound, remaining valid until March 31, 2029. This gives suppliers and hospitals the predictability they need to plan procurement and pricing — something that was often absent in the previous system.
The Bigger Picture: Where India’s Cancer Care Actually Stands
The duty exemption is one piece of a larger health budget that saw only modest growth. The Health Ministry received ₹1.05 lakh crore for FY 2026-27 — roughly six per cent higher than the previous year. Ayushman Bharat PM-JAY, the government’s flagship health insurance scheme, got ₹9,500 crore, barely above last year’s allocation.
The Budget also announced a ₹10,000 crore investment in the biopharma sector over five years, the creation of a nationwide network of 1,000 accredited clinical trial sites, and plans to set up three new National Institutes of Pharmaceutical Education and Research. These are infrastructure plays — the kind of moves that take years to translate into patient outcomes.
What stands out about the cancer drug exemption is its immediacy.
What’s Still Missing
Critics have already pointed out the limits. The duty removal doesn’t address the deeper structural problem: that most advanced cancer drugs in India are still priced well beyond what the average household can bear without insurance. Ayushman Bharat’s coverage caps and the gap between government hospital formularies and the drugs oncologists actually want to prescribe — those are problems that a customs duty tweak cannot solve.
There is also a question of whether manufacturers will actually pass the savings on. The Budget doesn’t mandate price reductions. Companies can choose to absorb the duty relief as improved margin instead. The National Pharmaceutical Pricing Authority has historically pushed back on this — in a previous round of duty cuts on life-saving drugs, the NPPA instructed manufacturers to pass savings directly to patients. Whether that happens here, and how aggressively it’s enforced, will determine whether this policy lands where it’s supposed to.
The Bottom Line
India’s Budget 2026 didn’t solve cancer. It didn’t even come close to solving the affordability crisis that defines how most Indians experience the disease. What it did — quietly, specifically, and with immediate effect — was remove a tax that had no business being on life-saving medicine in the first place.
For the patient on long-term breast cancer therapy, for the family scraping together ₹25 lakh for a one-time CAR-T treatment, for the rare disease patient importing medication through personal channels with no domestic alternative — this is a real change. Not a revolution. But a correction. And in a system that has been slow to make even those, it’s a step worth watching.
Sources: Union Budget 2026-27 documents presented in Parliament, February 1, 2026. Gazette notification on customs duty exemptions. Business Standard, The Hindu, The Week, DD News (News On Air), and TheHealthSite.com reporting. Drug indications verified against standard oncology references.
This article is for informational purposes. Treatment decisions should always be made in consultation with a qualified medical professional.